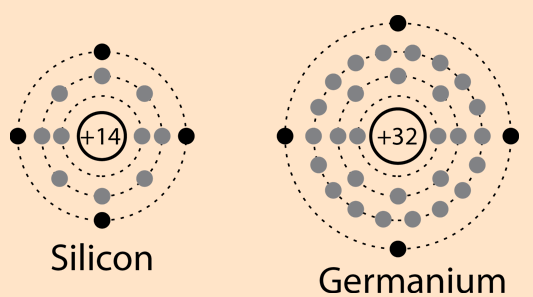
Silicon and Germanium
In fig the atomic structure of Silicon and Germanium are given we compared the atomic structures of Silicon and Germanium to each other. Silicon is used in semiconductor devices, diodes and integrated circuits. Both have four valence electrons. It is often say that it have valence electron in the 3rd shell and in the germanium the four valence electrons are in 4th shell. This shows that the silicons has less energy level and germanium has high energy level an extra additional energy is required to escape from the atom. At high temperature it makes the germanium more and more stable. That’s way the silicons is mostly used semiconductor.
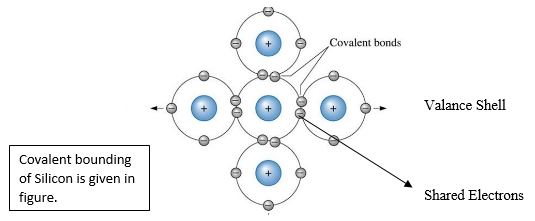
Covalent Bond
The figure that show how every silicon atom takes position itself to their adjacent silicons atom and make a silicons crystal. With their four valence electron of silicons donates an electron with every of its four neighbors. For each atom creates an eight shared valence electrons and produced the chemical stability. This type of sharing outer electron produced a covalent bonds that holds the atom together. Each valence electron is equally attracted by its adjacent atom. The impurities that are found in intrinsic silicons crystal in figure its shows clearly. The covalent bounding of germanium is same because it has also four valence electrons.