Semiconductors, Conductors, Insulators and Band Gap
Semiconductors
The semiconductor are those type of material that are between in conductors and insulators and it have ability to conduct the electrical current. The semiconductors are not good conductors neither good insulators they are in pure state. Antimony, arsenic, boron and polonium are the single element semiconductors. And most commonly used compound semiconductors are gallium, carbide, nitride and germanium. The mostly commonly used semiconductor is Silicon.
Conductors
The conductors are those type of material that allow the flow of charge are called conductors. The most common examples of conductor are aluminum, gold, silver and copper these all characterized by atoms there valence electron is very tightly bound to atom. The valance electron have free electrons. Therefore in conductive material that have free electrons at their valence shell.
Insulators
The insulators are those type of material that does not allow the flow of charge are called insulators. The compounds are the most good example of insulator a single element material that have very high resistivity there valance shell electrons are very tightly bound to the atom and they have very few free electrons in insulator. The most and common examples of insulators are plastic, glass and quartz.

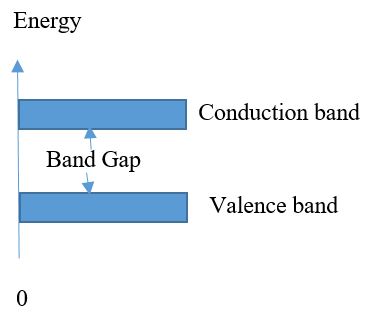
Band Gap
The band gap of energy level is represented by the valence shell of atom. And valence shell are very close to band. Enough energy is required by electrons. It leaves and become free electrons and exist in it is known as conduction band. The gap in energy between valence electron and conduction band is called energy gap or band gap. The energy that is present in valance electron it is in the form of sequence it has ability to jump from valance band to conduction band. The electrons freely move in conduction band.
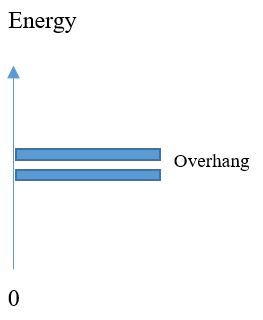
In fig that show the energy diagrams of Conductor, Insulator and Semiconductor. There exist a state in Insulators and Semiconductors there is no free electron is present. The electron jumps in certain conditions because there is no electron is present in this region in fig Band gap. For Insulators, the band gap cross when high voltages is applied and the break down condition is occur. In fig band gap diagram the insulators band gap is also given. In semiconductors the energy gap depends upon the semiconductor materials. In semiconductor the gap is little smaller then insulators and electron can easily jump from valence band to conduction band. The band gap of semiconductor is represented in fig band gap diagram. There is no gap is present in Conductors because the valence and conduction band overhang each other. The band gap is illustrated in fig band gap diagram.


